Ongoing Work
In no particular order:1. Aspects of the lysis-lysogeny decision in temperate bacteriophage (with Matt Scott, Shashi Thutupalli, Sine Svenningsen, Szabolcs Semsey and Vaibhhav Sinha)
2. The assembly of microbial communities (with Akshit Goyal, Kim Sneppen, Sergei Maslov and Szabolcs Semsey)
3. Lipid signalling in Drosophila phototransduction (with Raghu Padinjat and Rohit Suratekar)
4. The evolution of bistable switches via duplication (with Ian Dodd and Szabolcs Semsey)
5. The identification of transcription factor binding sites from ChIP seq data (with Vishaka Datta and Rahul Siddharthan)
6. The formation of morphogen gradients in plant embryos (with Madhu Venkadesan and Venkatesan Sundaresan)
7. Oscillations in the yeast metabolic cycle (with Sunil Laxman)
8. Entrainment and mode-hopping in NF-kB signalling (with Mathias Heltberg and Mogens Jensen)
9. Oscillations and travelling waves in somitogenesis (with Jonas Juul and Mogens Jensen)
10. Nonlinear coupling of wings and halteres in insect flight (with Akash Vardhan and Sanjay Sane)
11. Stochastic search processes with resets (with Kabir Husain)
12. Regulation exploiting correlated protein sysnthesis (with Amit Singh)
13. Interactions between CRISPR and RM systems in bacteria (with Aswin Seshasayee)
A Selection of Published Work
-
Molecular mechanisms of protein regulation
Hao et al. (2014) Road rules for traffic on dna – systematic analysis of transcriptional roadblocking in vivo, Nucl. Acids Res. 42, 8861– 8872.
 Genomic DNA is bound by many proteins that could potentially impede elongation of RNA polymerase, but the factors determining the magnitude of transcriptional roadblocking in vivo are poorly understood. Through systematic experiments and modeling, we analyse how roadblocking by the lac repressor in Escherichia coli cells is controlled by promoter firing rate, the concentration and affinity of the roadblocker protein, the transcription-coupled repair protein Mfd, and promoter–roadblock spacing.
Sneppen et al. (2010) Economy of operon formation: co-transcription minimizes shortfall in protein complexes. mBio 1(4):e00177-10.
Genomic DNA is bound by many proteins that could potentially impede elongation of RNA polymerase, but the factors determining the magnitude of transcriptional roadblocking in vivo are poorly understood. Through systematic experiments and modeling, we analyse how roadblocking by the lac repressor in Escherichia coli cells is controlled by promoter firing rate, the concentration and affinity of the roadblocker protein, the transcription-coupled repair protein Mfd, and promoter–roadblock spacing.
Sneppen et al. (2010) Economy of operon formation: co-transcription minimizes shortfall in protein complexes. mBio 1(4):e00177-10.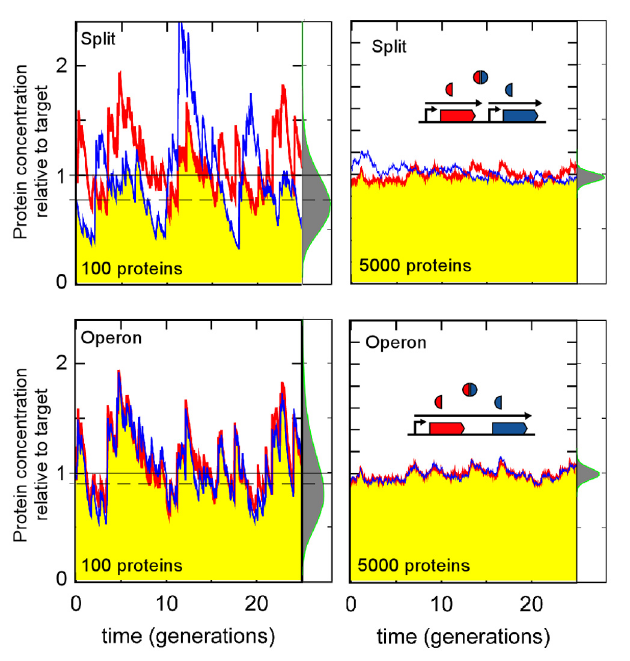 We show that there is a substantial economic gain for a cell to cotranscribe genes encoding protein complexes because it synchronizes
the fluctuations in the levels of the different components. This benefit is relatively large in small cells such as bacterial cells, in which
there are few mRNAs and proteins per cell, and is diminished in larger cells such as eukaryotic cells.
We show that there is a substantial economic gain for a cell to cotranscribe genes encoding protein complexes because it synchronizes
the fluctuations in the levels of the different components. This benefit is relatively large in small cells such as bacterial cells, in which
there are few mRNAs and proteins per cell, and is diminished in larger cells such as eukaryotic cells.
-
Life and death decisions in biological cells
Avlund et al. (2009) Why do phage play dice? J Virol. 83:11416-20. Phage lambda is among the simplest organisms that make a developmental decision. An infected bacterium goes either into the lytic state, where the phage particles rapidly replicate and eventually lyse the cell, or into a lysogenic state, where the phage goes dormant and replicates along with the cell. We argue that the phage are playing a "game" of minimizing the chance of extinction and that the shift from determinism to stochasticity is due to a shift from a single-player to a multiplayer game.Hunziker et al. (2010) Stress-specific response of the p53-Mdm2 feedback loop. BMC Syst Biol. 4:94. The p53 signalling pathway has hundreds of inputs and outputs. It can trigger cellular senescence, cell-cycle arrest and apoptosis in response to diverse stress conditions, including DNA damage, hypoxia and nutrient deprivation. Signals from all these inputs are channeled through a single node, the transcription factor p53. Yet, the pathway is flexible enough to produce different downstream gene expression patterns in response to different stresses. We show that even a simple negative feedback loop is capable of predicting the differences in p53 response to different stresses and single nucleotide polymorphisms.
-
Cellular communities
Heilmann et al. (2012) Coexistence of phage and bacteria on the boundary of self-organized refuges. Proc Natl Acad Sci U S A. 109:12828-33. Given that infection of a bacterium by a single virulent phage typically leads to the release of around 100 phage offspring, it’s a puzzle how virulent phage manage to coexist with bacteria without wiping them out. We used stochastic, spatial models of bacteria interacting with virulent phage to explore mechanisms involving spatial heterogeneity that enhance this coexistence. In particular, we look at the effect of density dependent mechanisms such as quroum sensing that can trigger the creation of bacterial refuges like biofilms.Heilmann et al. (2015) Why do bacteria regulate public goods by quorum sensing? Front Microbiol. 6:767. Use of a quorum-sensing system by bacterial populations makes most sense where production of a common good (e.g. virulence factors, biofilm constitutents, exoenzymes) in sufficient quantities may provide a substantial benefit to the entire population. We find that the shape of the benefit function (describing how the benefit increases, as the amount of common good increases) affects the way the quorum sensing system should be set up to maximize benefit. For a concave benefit function, it should turn on sharply and quickly at a critical density. Conversely, for a convex benefit function the quorum sensing system must turn on gradually as density increases. A sharp turn on is in fact what is observed in most experimental situations, and is caused by positive feedback loops in the quorum system regulatory network.
-
Oscillations & entrainment
Jensen et al. (2012) Inducing phase-locking and chaos in cellular oscillators by modulating the driving stimuli. FEBS Lett. 586:1664-8. Inflammatory responses in eukaryotic cells are often associated with oscillations in the nuclear-cytoplasmic translocation of the transcription factor NF-kB. We used a mathematical model to show that an oscillatory external stimulus can synchronize the NF-kB oscillations into states where the ratios of the internal to external frequency are close to rational numbers. We predict a specific response diagram of the TNF-driven NF-kB system which exhibits bands of synchronization known as “Arnold tongues". Our model also suggests that when the amplitude of the external stimulus exceeds a certain threshold there is the possibility of coexistence of multiple different synchronized states and eventually chaotic dynamics of the nuclear NF-kB concentration. This could be used as a way of externally controlling immune response, DNA repair and apoptotic pathways.Mengel et al. (2010) Modeling oscillatory control in NF-κB, p53 and Wnt signaling. Curr Opin Genet Dev. 20:656-64. In the first part of this article, we provide a brief overview of simple deterministic models of oscillations. In particular, we explain the mechanism of saturated degradation that has been used to model oscillations in the NF-κB, p53 and Wnt systems. The second part deals with the potential physiological role of oscillations. We use the simple models described earlier to explore whether oscillatory signals can encode more information than steady-state signals. We then discuss a few simple genetic circuits that could decode information stored in the average, amplitude or frequency of oscillations.
-
Bacterial metabolism
Semsey et al. (2013) The effect of LacI autoregulation on the performance of the lactose utilization system in Escherichia coli. Nucleic Acids Res. 41:6381-90. The lactose operon of Escherichia coli is a paradigm system for quantitative understanding of gene regulation in prokaryotes. Yet, none of the many mathematical models built so far to study the dynamics of this system considered the fact that the Lac repressor regulates its own transcription by forming a transcriptional roadblock at the O3 operator site. Previously, it has been argued that negative autoregulation can significantly reduce noise as well as increase the speed of response. We show that the particular molecular mechanism, a transcriptional roadblock, used to achieve self-repression in the lac system does neither. Instead, LacI autoregulation balances two opposing states, one that allows quicker response to smaller pulses of external lactose, and the other that minimizes production costs in the absence of lactose.Horvath et al. (2010) Timing of gene transcription in the galactose utilization system of escherichia coli, J. Biol. Chem. 285, 38062–38068. In the natural environment, bacterial cells have to adjust their metabolism to alterations in the availability of food sources. The order and timing of gene expression are crucial in these situations to produce an appropriate response. We used the galactose regulation in Escherichia coli as a model system for understanding how cells integrate information about food availability and cAMP levels to adjust the timing and intensity of gene expression. We simulated the feast-famine cycle of bacterial growth by diluting stationary phase cells in fresh medium containing galactose as the sole carbon source. We followed the activities of six promoters of the galactose system as cells grew on and ran out of galactose. We found that the cell responds to a decreasing external galactose level by increasing the internal galactose level, which is achieved by limiting galactose metabolism and increasing the expression of transporters. We show that the cell alters gene expression based primarily on the current state of the cell and not on monitoring the level of extracellular galactose in real time. Some decisions have longer term effects; therefore, the current state does subtly encode the history of food availability. In summary, our measurements of timing of gene expression in the galactose system suggest that the system has evolved to respond to environments where future galactose levels are unpredictable rather than regular feast and famine cycles.